An important aspect for the success of bioelectronics is reducing the immune response of an organism to the electronic device. An ideal implant is biologically inert and does not activate a response from the host. If the bioelectronic device causes an immunological response, the device may become encapsulated within fibrous tissue which can disrupt the interface between the neural tissue and device. The biocompatibility of a polymer is tested using many different methods ranging from cytotoxicity and sensitisation assays to hemocompatibility and genotoxicity. Therefore, biocompatibility is an important requirement that needs to be met for a polymer to become suitable.
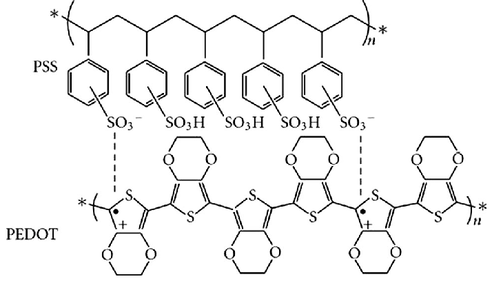
The range of current conjugated polymers used in bioelectronics is quite narrow with the most common ones being polypyrrole (Ppy), polyaniline (PANI) and polythiophene derivatives such as poly(3,4-ethylenedioxythiophene) doped with poly(styrenesulfonate) (PEDOT:PSS).
The range of current conjugated polymers used in bioelectronics is quite narrow with the most common ones being polypyrrole (Ppy), polyaniline (PANI) and polythiophene derivatives such as poly(3,4-ethylenedioxythiophene) doped with poly(styrenesulfonate) (PEDOT:PSS).
Early designs of conjugated polymers were based on polypyrrole and polyanilines both synthesised by the oxidation of pyrrole and aniline respectively with the conductive form of Ppy formed via oxidative p-doping of the polymer.
Polypyrrole coatings have a great thermal stability and can be used in carbon composites. They are used in micro actuators, solid electrolytic capacitors and polymeric batteries whereas Polyanilines are used in solar batteries, as a catalyst and in transparent electrostatic coatings.
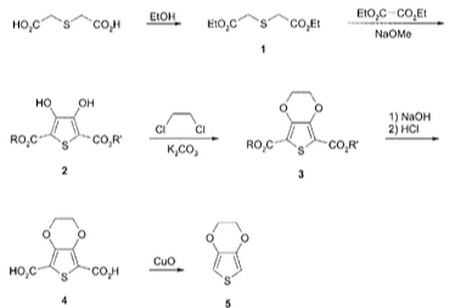
However, both these polymers were found to be unstable, easily overoxidized and insoluble in organic solvents which limits their use in bioelectronics due to the abundance of oxidative species in aqueous biological environments. Also, the ability for the polymer to remain stable during overoxidation is an important requirement in supporting changes in the charge density. These limitations have propelled polymers based on the 3,4-ethylenedioxythiophene (EDOT) repeat unit as shown below.
Polypyrrole coatings have a great thermal stability and can be used in carbon composites. They are used in micro actuators, solid electrolytic capacitors and polymeric batteries whereas Polyanilines are used in solar batteries, as a catalyst and in transparent electrostatic coatings.
However, both these polymers were found to be unstable, easily overoxidized and insoluble in organic solvents which limits their use in bioelectronics due to the abundance of oxidative species in aqueous biological environments. Also, the ability for the polymer to remain stable during overoxidation is an important requirement in supporting changes in the charge density. These limitations have propelled polymers based on the 3,4-ethylenedioxythiophene (EDOT) repeat unit as shown below.
As a result, PEDOT:PSS was formed by oxidising EDOT monomers to form the PEDOT polymer structure and inserting a negative PSS dopant to counteract the positive charge of the PEDOT structure. It is the most widely used polymer for commercial applications due to its water solubility, high transparency and good ductility. The conductive and mechanical properties of PEDOT:PSS can be enhanced using different methods such as using solvents and altering the crystallinity of the polymer which improves its efficiency in bioelectronics.
To improve the biocompatibility, biopolymers can be used as a dopant instead of PSS which can overcome limitations for specific applications. Popular biopolymer dopants include guar gum, DNA, dextran sulfate and hyaluronic acid. In addition, further solvents or secondary dopants e.g ethylene glycol can be added to increase conductivity.
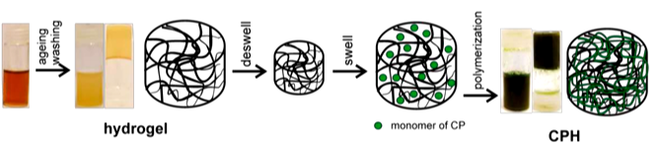
Recent advancements in bioelectronics has increased the seamless merging between biology and electronics through the introduction of hydrogels due to their resemblance to biological tissues. Hydrogels are 3D networks of hydrophilic polymers that can hold large amounts of water whilst maintaining its structure due to the cross-linking of the polymer chain. Conductive polymers hydrogels are used due to its electrical properties and the high-water content which provides a wet and ion-rich environment. PEDOT:PSS based hydrogels are commonly used and can be enhanced using the solvent dimethyl sulfoxide (DMSO).
Recent advancements in bioelectronics has increased the seamless merging between biology and electronics through the introduction of hydrogels due to their resemblance to biological tissues. Hydrogels are 3D networks of hydrophilic polymers that can hold large amounts of water whilst maintaining its structure due to the cross-linking of the polymer chain. Conductive polymers hydrogels are used due to its electrical properties and the high-water content which provides a wet and ion-rich environment. PEDOT:PSS based hydrogels are commonly used and can be enhanced using the solvent dimethyl sulfoxide (DMSO).
Research is currently being conducted in improving all these polymers and discovering different ways to improve the mechanical and conductive properties without comprising its biocompatibility.